
FDA Clears New Covid Vaccines with Eligibility Limits Under RFK Jr.
 :
| Updated On: 28-Aug-2025 @ 12:27 pm
:
| Updated On: 28-Aug-2025 @ 12:27 pmSHARE
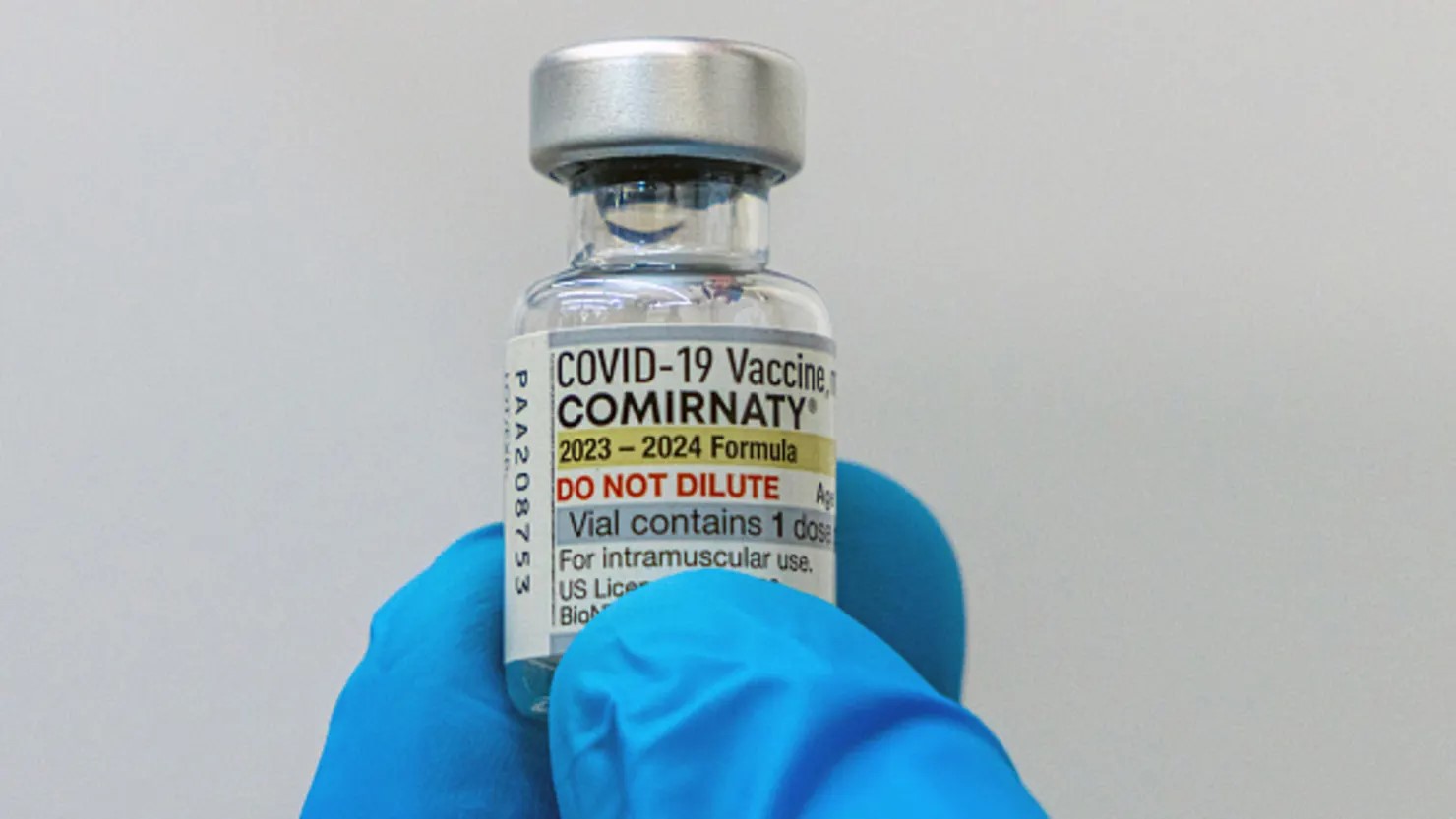
The FDA approved new Covid vaccines in the U.S. but restricted access to high-risk groups, including adults 65+ and younger people with underlying conditions. This ends broad emergency use authorizations, limiting availability for healthy adults and children under 5. Moderna, Pfizer, and Novavax shots remain authorized for specific age and risk groups. Critics, including the American Academy of Pediatrics, called the move troubling, warning it reduces protection for infants, toddlers, and pregnant women. HHS Secretary Robert F. Kennedy Jr., a vaccine skeptic, framed the decision as a return to “science, safety, and common sense,” sparking debate over access and policy.